Measuring Creatine and Creatinine in Finished Products: Why Compendial Methods Fall Short
April 20, 2026

Overview
Creatine is one of the most widely used ingredients in sports nutrition, and one of the most poorly tested in finished product form.
Most analytical methods in commercial use today were designed for one of two things: assessing the purity of pharmaceutical-grade raw materials, or measuring creatine and creatinine in biological matrices like urine. Neither was built to handle the complex, multi-ingredient formulations that define today's supplement market — gummies, stick packs, drink powders, protein blends, and multi-component pre-workouts.
The result is a blind spot. Methods that perform well on pure creatine powder can produce misleading data when applied to finished goods — overstating creatine content, understating degradation, or both. For brands building label claims, stability programs, and quality systems around that data, the consequences are real.
This paper explains where existing methods break down, what a purpose-built approach looks like, and why the distinction matters for anyone formulating with creatine.
Background: The Analytical Challenge of Creatine in Complex Matrices
Creatine and creatinine are small, polar, water-soluble molecules. On most chromatographic systems, they elute early and fast — occupying a retention window that is crowded with other compounds commonly found in dietary supplement formulations.
In a pure creatine monohydrate powder, this isn't a problem. The matrix is clean, interferences are minimal, and even a method with modest selectivity can produce an accurate result. But finished dietary supplements are a different analytical environment entirely.
Consider what a typical creatine-containing finished product might include alongside the active ingredient: free amino acids like glycine, taurine, or beta-alanine in a pre-workout blend; organic acids like citric acid and malic acid in a gummy or flavored drink mix; B-vitamins such as niacinamide or pyridoxine in a multivitamin-sport hybrid; sweeteners, flavoring agents, and other excipients that vary by product format. Many of these compounds are polar, UV-absorbing, and chromatographically similar to creatine and creatinine under standard reversed-phase or normal-phase conditions. In a multi-ingredient formulation, the risk of co-elution — where an interfering compound is captured within the creatine or creatinine peak and quantified as part of the result — becomes a real analytical concern.
The consequence is straightforward: if your method can't resolve creatine from these matrix components, your reported value may not reflect what's actually there. In high-dose creatine products where samples are significantly diluted prior to analysis, co-eluting interferences become proportionally smaller relative to the creatine signal — which can make the problem easy to overlook, but doesn't eliminate it. And for creatinine, which may be present at trace levels in a well-preserved product, even modest interference can meaningfully distort the result.
This is the core analytical challenge that any creatine method intended for finished products needs to solve: sufficient selectivity to resolve the target analytes from the complex polar background of a real supplement matrix.
Where Existing Methods Fall Short
Measuring creatine and creatinine is not new. Both analytes appear in the USP creatine monograph, and numerous commercial and academic methods exist for their determination. The issue isn't whether these analytes can be measured — it's whether the methods in common use produce reliable data across the range of matrices that matter in today's supplement market.
The USP monograph was designed for raw material purity and process-related impurity screening. The column chemistries and sample preparation approach reflect that scope — they provide adequate separation of creatine from its process impurities in a clean, single-ingredient matrix, but were never intended to resolve creatine and creatinine from the broader range of polar compounds present in complex finished products. Creatinine itself is measured only as a process-related impurity against a limit threshold — not as a quantitative determination with the dynamic range needed to monitor degradation in a finished product where creatine may be present at 100- to 1000-fold higher concentrations. The sample diluent is water, with no pH buffering — adequate for a pure powder, but potentially problematic for acidic finished-product matrices where uncontrolled pH can drive artifactual creatine-to-creatinine conversion during preparation.
Industry methods outside of regulatory frameworks present a different set of limitations. Most are designed for biological matrices like urine, or for simple single-ingredient powders. A standard C18 reversed-phase method works well enough when the matrix is clean — but creatine and creatinine are poorly retained on C18 due to their high polarity, eluting early with limited separation from other polar matrix components. In a multi-ingredient finished product, that lack of retention translates directly to co-elution risk.
The gap isn't in whether creatine and creatinine can be measured. It's in whether the method is robust enough to produce trustworthy data across the full range of product formats the supplement industry actually makes.
The Ion-Pairing Approach: Selectivity Across Complex Matrices
The core technical decision in our method is the use of an ion-pairing reagent in conjunction with a C18 column. This combination solves the fundamental selectivity problem described above: creatine and creatinine are polar, water-soluble compounds that elute very quickly on a standard reversed-phase system — too quickly to be reliably separated from matrix interferences in complex formulations.
The ion-pairing reagent dramatically improves chromatographic retention of these analytes through mechanisms that likely involve both ion-pair complex formation in the mobile phase and dynamic surface modification of the stationary phase. The result is strong, consistent retention that gives the chromatographic system the resolving power it needs to separate creatine and creatinine from other polar compounds — amino acids, organic acids, vitamins — present in finished supplement matrices.
This is what makes the method work for both pure ingredients and complex finished products in the same analytical framework. A raw material creatine powder and a multi-ingredient gummy can be analyzed on the same platform, with the same confidence in selectivity, without changing columns or reconfiguring the system.
Two Complementary Mechanisms
The retention behavior of ion-pairing chromatography is explained by two complementary models, and in practice, both likely contribute.
1. Ion-Pair Formation Model
The ion-pairing reagent forms a discrete complex with the charged analyte in the mobile phase. This complex carries reduced overall charge and increased lipophilicity, allowing it to partition into the non-polar stationary phase based on its altered properties — effectively pre-neutralizing the analyte's charge before it reaches the column surface.

2. Dynamic Ion-Exchange Model
The ion-pairing reagent adsorbs onto the stationary phase surface through its lipophilic alkyl chain, creating transient ion-exchange sites. The charged analyte then interacts directly with these dynamically formed sites, producing retention through a mechanism that is functionally equivalent to ion-exchange chromatography — but on a standard reversed-phase column.
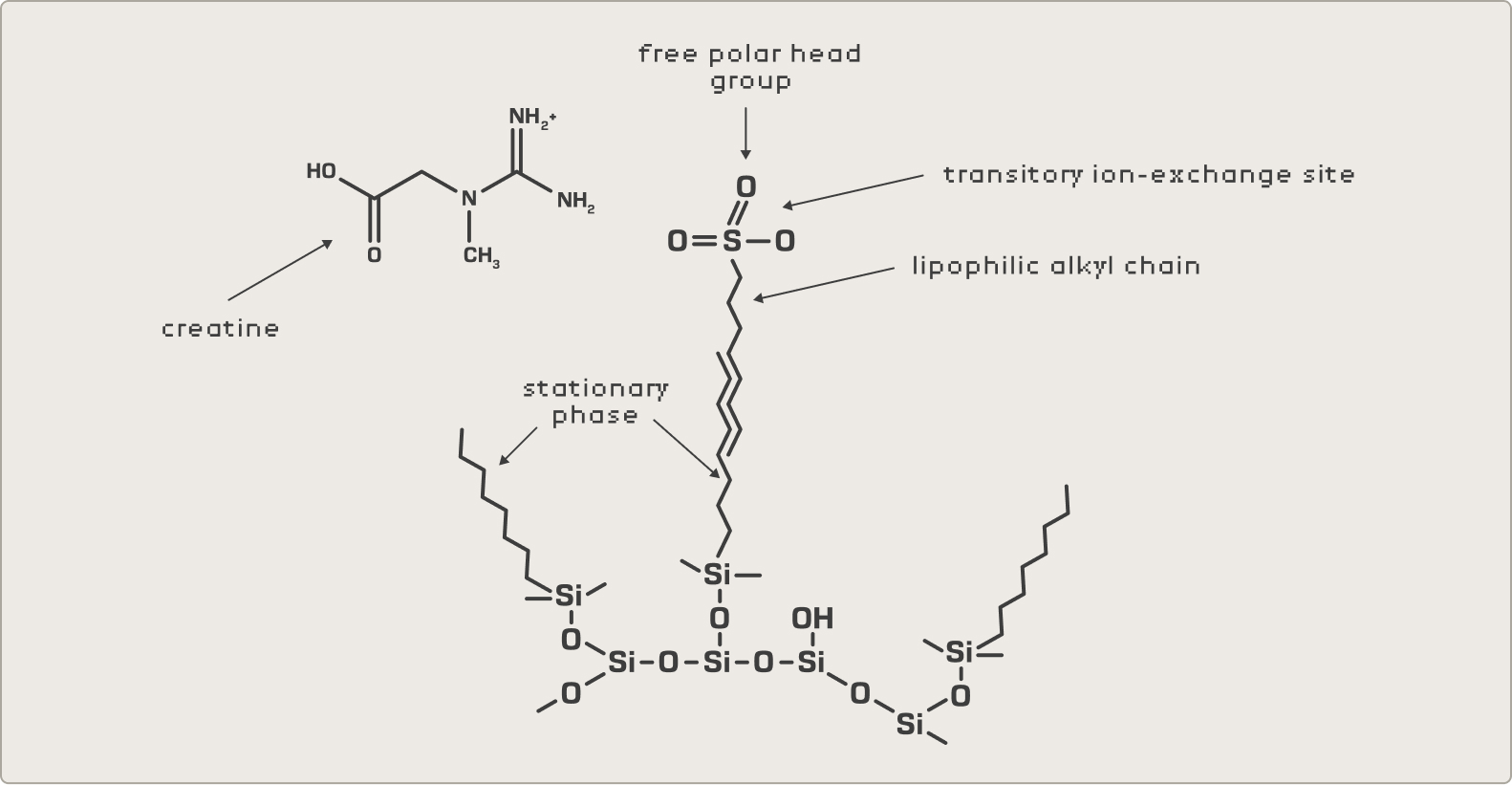
The practical significance is that this chemistry gives us the selectivity advantages of ion-exchange with the flexibility and robustness of a reversed-phase platform — a combination that is particularly well-suited to the diverse matrix types encountered in dietary supplement testing.
A Critical Detail: pH Control During Sample Preparation
One of the most important — and most easily overlooked — aspects of creatine analysis is what happens to the sample before it reaches the instrument.
Creatine undergoes spontaneous cyclization to form creatinine — a lactam byproduct with no ergogenic benefit whose accumulation signals active ingredient loss. This conversion is pH-dependent: both strongly acidic and strongly alkaline conditions accelerate the reaction. Under conditions of elevated temperature or inadequate storage, this degradation occurs in the product itself — and because creatinine formation occurs at the direct expense of creatine content, a product may underdeliver on label claims without any visible signs of degradation.
But this same conversion can also happen in the lab. If sample preparation is not carefully controlled, creatinine can be generated artifactually — producing a result that overstates degradation relative to what was actually present in the product.
This is particularly relevant for finished-goods matrices. Gummies, for example, typically contain citric acid and are naturally acidic. Without pH intervention, dissolving a gummy into an aqueous solution creates conditions that actively drive the creatine-to-creatinine conversion during preparation — before the sample is even injected. As noted above, the USP method specifies plain water as the sample diluent, with no provision for pH control — an approach that is adequate for a pure powder but potentially problematic for acidic finished-product matrices.
Our method uses a specialized buffer system at a concentration and pH optimized to neutralize the acidity of common finished-goods matrices and maintain sample stability throughout the entire analytical sequence. We specifically evaluated and moved away from volatile buffer systems, which drift in pH over time — a meaningful concern when samples may sit in an autosampler queue overnight before injection.
Our internal stability work confirmed the stakes: uncontrolled pH during sample preparation produces measurable creatinine formation within hours, even at room temperature. The buffering approach we selected maintains pH stability indefinitely at room temperature, ensuring that the result reflects the product — not the sample prep.
Robustness Across Matrices
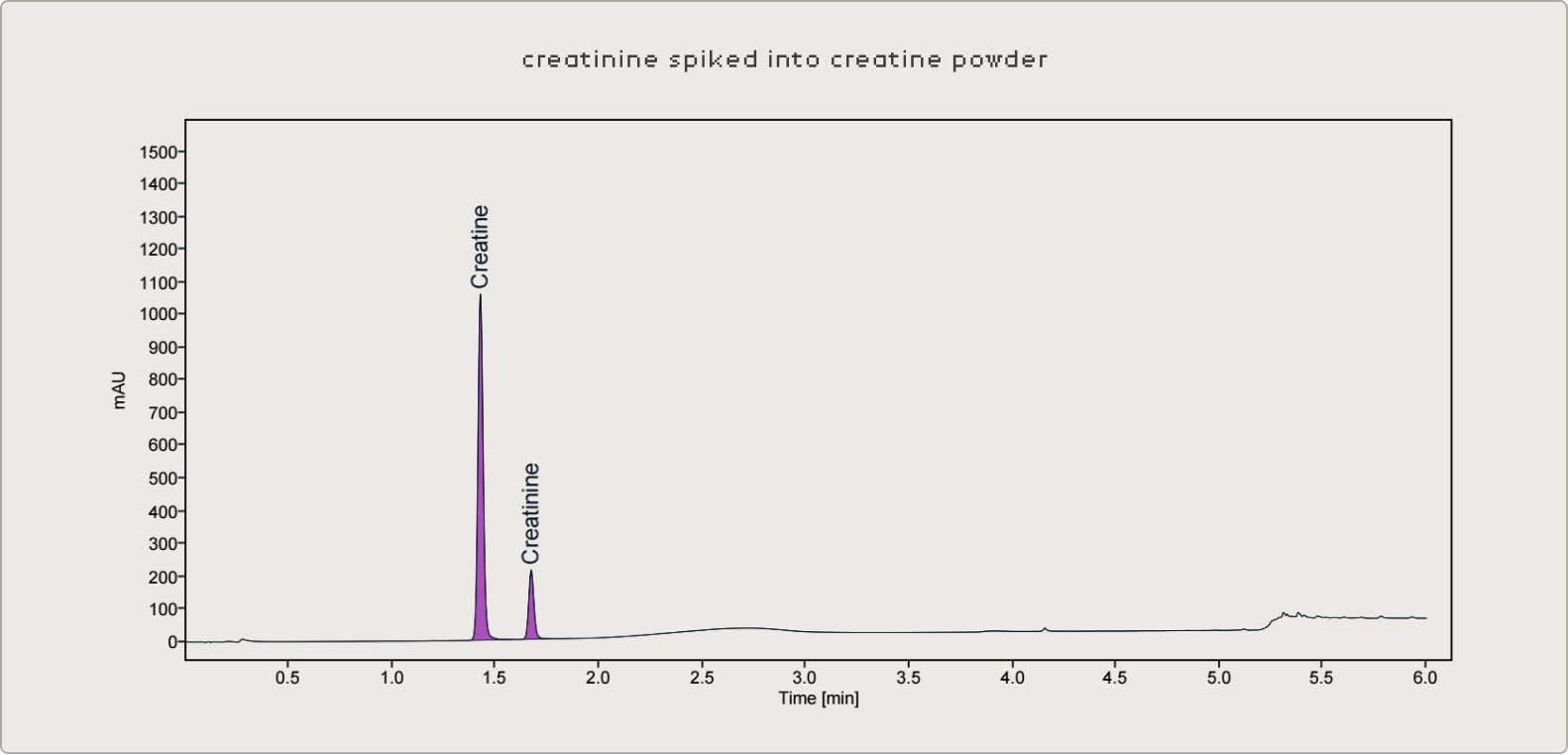
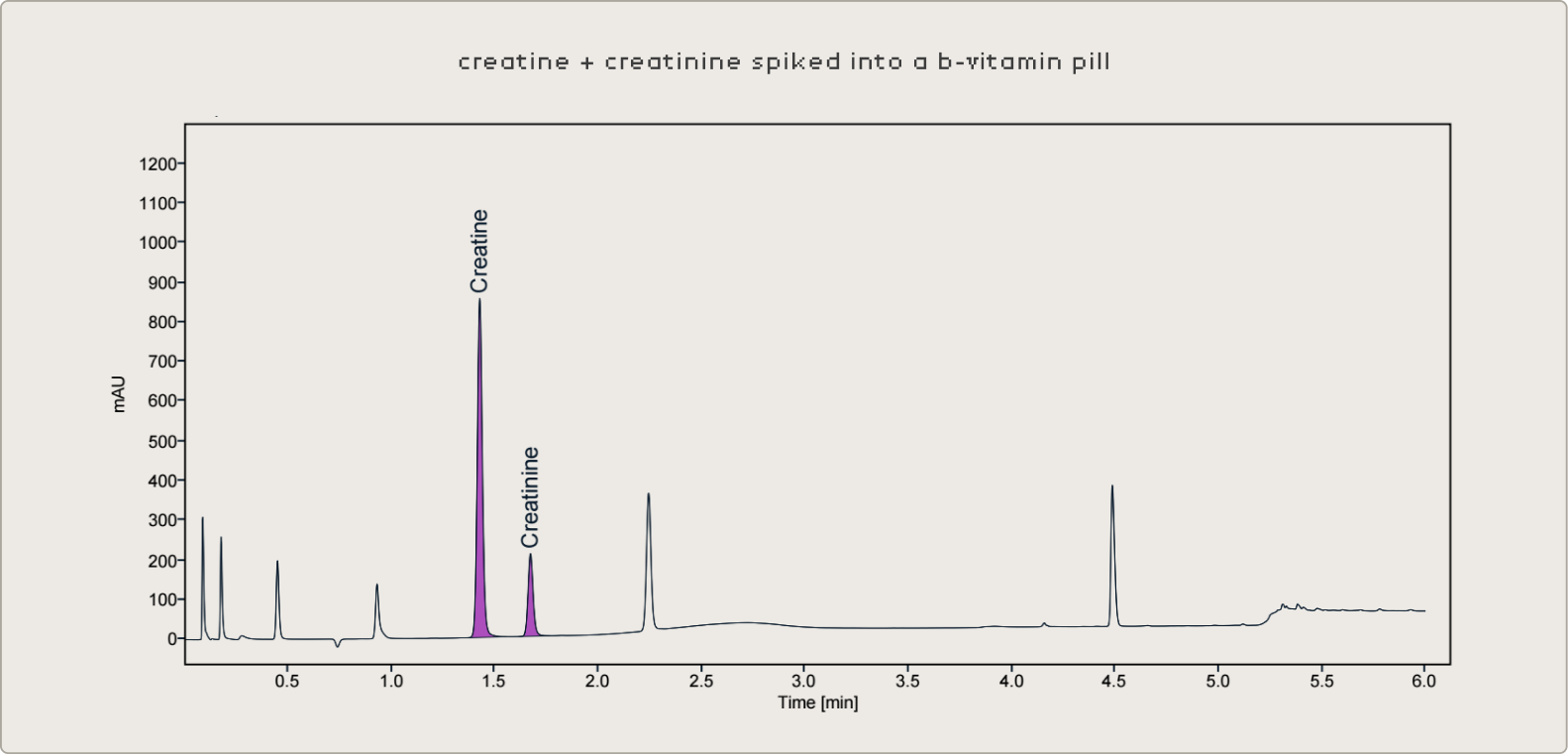
To validate the method's performance across the range of product formats seen in the supplement industry, known concentrations of creatine and creatinine were spiked into a panel of dietary supplement products not originally containing creatine — including gummies, multi-ingredient drink powders containing L-theanine and other amino acids, and B-vitamin gummies. Samples were prepared at multiple dilution factors spanning the calibration range to confirm that accuracy is consistent regardless of where in the curve the result falls.
This was particularly important for the simultaneous quantification of both analytes. In a well-preserved product, creatinine may be present at very low concentrations relative to creatine. The method needs to maintain a low limit of quantification for creatinine while accurately measuring creatine at much higher concentrations — all in the same injection. The dilution and buffering strategy was optimized with this dynamic range challenge in mind.
Who Should Be Testing
This method is designed for brands formulating with creatine who need to verify label claims and monitor product stability, ingredient suppliers demonstrating raw material purity and quality to their customers, contract manufacturers monitoring process conditions and their impact on ingredient integrity, and quality and regulatory teams building out stability protocols or responding to out-of-spec investigations.

get started
complete this form to connect with a cambium analytica team member





